Smelter acid will be more than 25 of world sulphuric acid production com-pared to some 18 in 1991. The contact process involves three main steps.
2 4 Acids And Bases Medicine Libretexts
NaCl H2SO4 NaHSO4 HCl 2 NaCl NaHSO4 Na2SO4 HCl 3 2NaCl H2SO4 Na2SO4 2HCl 4 3.
H acid manufacturing process pdf. Elevated process temperatures if the concentration of the acid continuously increased during bath lifetime. First step is oxidation of sulphur to sulphur dioxide. Production and Process Controls 21 CFR 211 Subpart F 4 A.
Boiling point temperatures of ortho -phosphoric acid vs. The ionisation of sulfuric acid is exothermic releasing lots of heat. H2 Cl2 2HCl 1 2.
High-purity hydrofluoric HF acid begins with the mixing and chemical interaction of sulfuric acid and dry fluorspar CF2. A method to produce the Sulfuric acid in lead lined chambers. Thermal process phosphoric acid manufacture as shown schematically in Figure 12 involves three major steps.
The HF gas is condensed then distilled to form anhydrous hydrofluoric acid. Description of Nitric acid manufacturing process Sameer Pandey1 National Fertilizers Limited Naya Nanagal Ropar Punjab140126 Abstract. Direct Oxidation Process When the H 2 S concentration is below about 15 in the acid gas the direct oxidation version of the Modified Claus Process may be used.
1 combustion 2 hydration and 3 demisting. Liquid juice worm fertilizers húmus líquido. Lead Chamber Process Contact Process.
Filtration Washing 5. Second step is catalytic oxidation of sulphur dioxide to sulphur trioxide. H 2 SO 4 aq Haq HSO 4aq heat Sulfuric acid dissociates in two steps.
Rather than using a burner to combust H 2 S to form SO 2 the direct oxidation process catalytically reacts oxygen with H 2 S by mixing the air and acid gas upstream of a catalytic. The most important uses of sulfuric acid include. This process has been abandoned because of the amount of energy which is needed.
Equipment 21 CFR 211 Subpart D 3 IV. The pure compound is a pungent-smelling colourless gas that polymerises spontaneously into paraformaldehyde refer to section Forms below hence it is stored as an aqueous. H-acid 1-amino-8-naphthol-3 6-disulfonic acid was selected as target compound to evaluate the performance of this novel process.
Description of nitric acid manufacturing process 1. Disinfectant formulation and manufacturing process 3. Sulphuric Acid Manufacturing Process.
The processes that use phosphated minerals which are decomposed with an acid are known as. Formulations and production process 61 citric acid based hemodialysis machines disinfectant formulation and manufacturing process 1. The result of mixing those in a rotary kiln is HF gas.
Adhesives formulations encyclopedia text-book pdf. Filtration Washing 7. Processes with different raw materials are used in the manufacture of phosphoric acid.
The removal of H-acid and TOC total organic carbon for H-acid. Notice that sulfuric acid is a strong acid in its first dissociation but the HSO 4 ion is a weak acid and only dissociates slightly. Third step is hydration of sulphur trioxide to form Sulphuric Acid.
184 gcm³ Molar mass. Minivadia Niraj Patel Nishit J. Components and Product Containers 21 CFR 211 Subpart E 4 V.
Some ingredients used in. Critical Manufacturing Steps 21 CFR 211101 4. 2 gas that was passed over catalytic be ds to produce sulfur trioxide SO.
Lead-acid batteries Ore processing Fertilizer manufacturing Oil refining Wastewater processing Chemical synthesis Production Sulphuric Acid can be manufactured by two processes. RAW MATERIAL CHEMICALS Raw Manufacturing. In the manufacturing process of H-Acid following Steps are involved.
It was absorbed through distil led water to produce sulfuric acid H. Typical steps and reactions of the lead chamber process are as follows Lloyd 2011. 862 Process Description1-4 Hydrochloric acid can be produced by 1 of the 5 following processes.
Of Chemical Engineering Parul Institute of technology Vadodara India Abstract The primary objective is to design a chemical plant for the production of nitric acid by ammonia oxidation process. 337 C Importance One of the most important of all chemicals. Sulfuric acid Oleum Naphthalene.
H 2 SO 4 aq Haq H. Thermal Process Acid Production Raw materials for the production of phosphoric acid by the thermal process are elemental yellow phosphorus air and water. 98079 gmol Boiling point.
Nitric acid is a very valuable chemical and finds intensive use in polymer industry as an oxidizing agent and in producing other chemicals. Reaction of metallic chlorides particularly sodium chloride NaCl with sulfuric acid H2SO4 or a hydrogen sulfate. Good Manufacturing Practices in Over-the-Counter Drug Product Manufacturing 3 I.
Formaldehyde f ə r ˈ m æ l d ə h aɪ d fer-mal-duh-hahyd also f ɔːr ˈ m æ l d ə h aɪ d for- systematic name methanal is a naturally occurring organic compound with the formula CH 2 O HCHO. Patel Rishit Patel Dept. 12 Sulfuric acid was manufactured by melting and burning raw sulfur to produce sulfur dioxide SO.
Therefore continuous r eplenishment of H 2O is required to maintain the etch rate and selectivity at a given process temperature. Process Analysis and Plant Design for Manufacturing of Nitric Acid Riyaz S. Whereas in 1991 smelter acid production amounted to 2798 millions tonnes it is calculated that the output in the following decade will have grown to reach 4497 millions tonnes in 2001.
When absorbed through distilled water 60 strength nitric acid HNO. Step 1 - Burning of KNO 3 potassium nitrate. The process is known as thermal when the raw material is elemental phosphorus.

O Level Chemistry Sulfur Chemistry O Levels Process

Images Of Power Plant Process Flow Diagram Diagrams Process Flow Diagram Process Flow Cycle Process

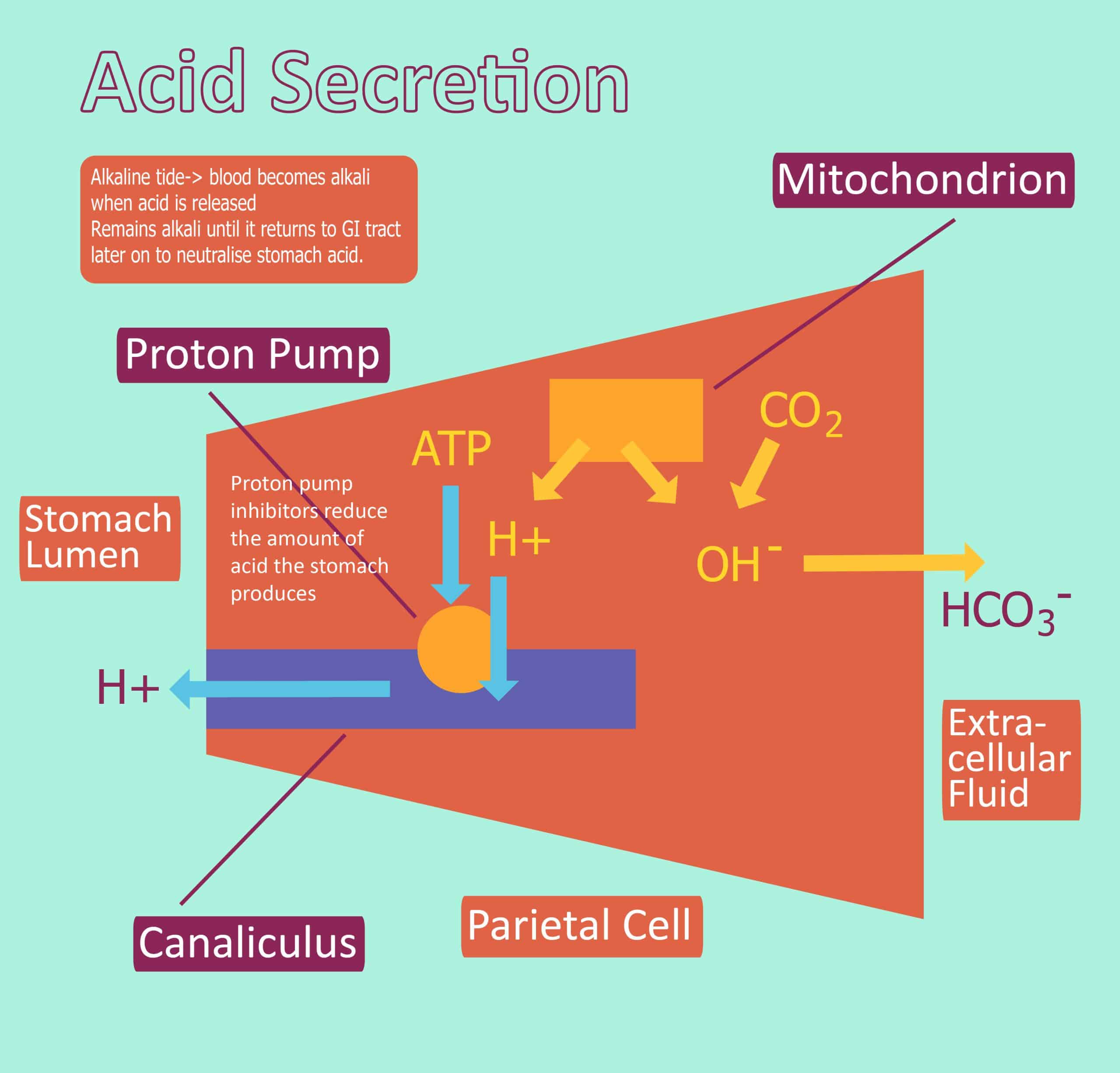
Acid Production Hydrochloric Acid Regulation Teachmephysiology

92 Questions With Answers In Process Engineering Science Topic
