PAT Applied in Biopharmaceutical. Save on PAT Applied in Biopharmaceutical Process Development And Manufacturing An Enabling Tool for Quality-by-Design by Cenk Undey.

Process Analytical Technology Achieving Process Understanding The Foundation Of A Strategic Pat Program Pharmaceutical Manufacturing
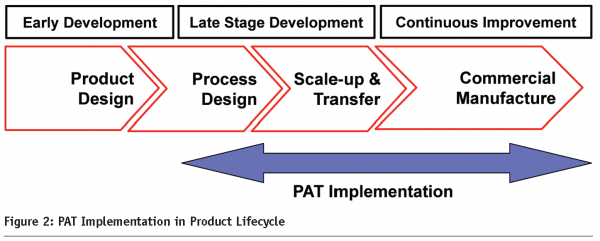
Process to achieve the desired product attributes process analytical technology PAT is an important tool for QbDPAT tools are routinely applied to develop a greater understanding of the process design space under a Quality-by-Design QbD framework.
Pat applied in biopharmaceutical process development and manufacturing. Process Scale Bioseparations for the Biopharmaceutical Industry brings together scientific principles empirical approaches and practical considerations for designing industrial downstream bioprocesses for various classes of biomolecules. PAT Applied in Biopharmaceutical Process Development and Manufacturing An Enabling Tool for Quality-by-Design. While for biopharmaceutical manufacturers to evaluate their need and ability to implement new technologies.
Shop your textbooks from ZookalAU today. Making predictions about the key process end points such as process performance indicators or quality attributes using a process model offers the unique advantages of process improvement and optimisation and helps give insights into variability. Process Analyzer Technology Biotechnology processes present technical challenges different from those in the pharmaceutical industry.
Predictive monitoring is a key feature of biopharmaceutical manufacturing. QbD assures the quality of a pharmaceutical product through scientific development and risk management tools and will eventually enable real-time release regardless of the formulation type. Skickas inom 10-15 vardagar.
PAT Applied in Biopharmaceutical Process Development and Manufacturing covers technological advances in measurement sciences data acquisition monitoring and control. Save up to 80 by choosing the eTextbook option for ISBN. It is also very possible that the final version of the PATguidance will apply to biopharmaceutical manufacturing.
Technical leaders present real-life case studies in areas including measuring and monitoring raw materials cell culture purification and cleaning and lyophilization processes via advanced PAT. The use of PAT tools helps enable the development of robust processes. PAT Applied in Biopharmaceutical Process Development And Manufacturing An Enabling Tool for Quality-by-Design 1st Edition by Cenk Undey and Publisher routledge.
PAT Applied in Biopharmaceutical Process Development and Manufacturing covers technological advances in measurement sciences data acquisition monitoring and control. Pat applied in biopharmaceutical process development and manufacturing an enabling tool for quality by design biotechnology and bioprocessing is available in our book collection an online access to it is set as public so you can get it instantly. Ensure batch-to-batch consistency from product development to production.
PAT Applied in Biopharmaceutical Process Development and Manufacturing covers technological advances in measurement sciences data acquisition monitoring and control. Technical leaders present real-life case studies in areas including measuring and monitoring raw materials cell culture purification and cleaning and lyophilization processes via advanced PAT. Ad Analytics to support pharmaceutical RD scale-up and production.
The print version of this textbook is ISBN. The methods and strategies described are essential learning for every scientist engineer or manager in the biopharmaceutical and vaccines industry. Bioseparations-also called downstream processing- can be a key source of competitive advantageto biopharmaceutical developers.
Pat Applied In Biopharmaceutical Process Development And Manufacturing An Enabling Tool For Quality By Design Biotechnology And Bioprocessing Recognizing the habit ways to acquire this book pat applied in biopharmaceutical process development and manufacturing an enabling tool for quality by design biotechnology and bioprocessing is additionally useful. Development Design and Implementation of Manufacturing Processes covers bioprocessing from cell line development to bulk drug substances. This first chapter introduces the concept of quality-by-design QbD and its role in pharmaceutical product development.
Technical leaders present real-life case studies in areas including measuring and monitoring raw materials cell culture purification and cleaning and lyophilization processes via advanced PAT. Process analytical technology PAT has been gaining momentum in the biotech community due to the potential for continuous real-time quality assurance resulting in improved operational control and. As with all of pharmaceutical production the regulatory environment for the production of therapeutics has been changing as a direct result of the US FDA-initiated Quality by Design QbD guidelines.
PAT Applied in Biopharmaceutical Process Development and Manufacturing covers technological advances in measurement sciences data acquisition monitoring and control. PAT Applied in Biopharmaceutical Process Development And Manufacturing book An Enabling Tool for Quality-by-Design Edited By Cenk Undey Duncan Low Jose C.
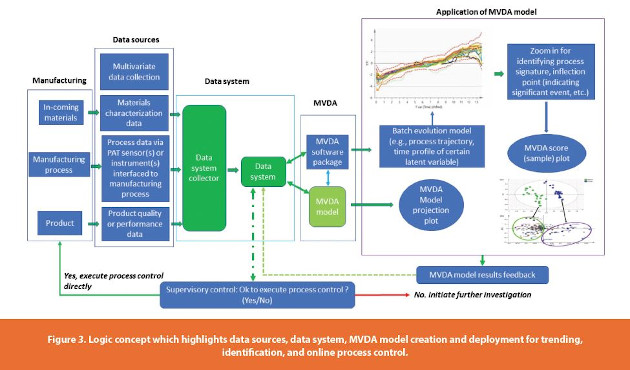
Integration Of Pat And Data Systems For Manufacturing Process Control And Quality Improvement A Brief Review Of Past Developments Current Status And Certain Future Considerations American Pharmaceutical Review The Review
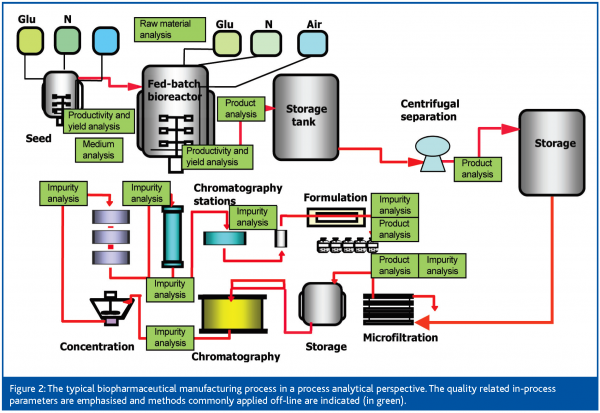
The Role Of Pat In Biotechnology European Pharmaceutical Review
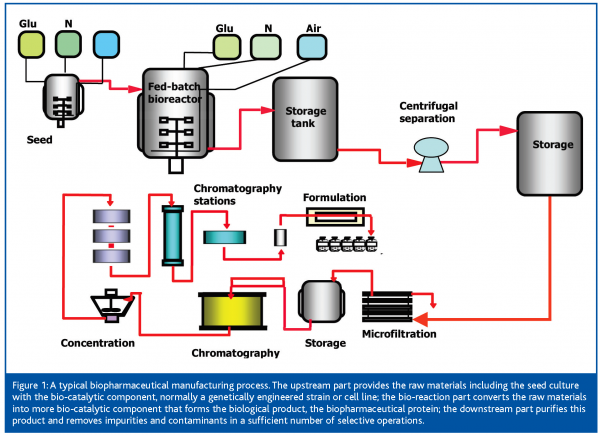
The Role Of Pat In Biotechnology European Pharmaceutical Review

Successful Pat Implementation In Pharmaceutical Manufacturing
